On January 25, Associate Professor Guan Ji-Song’s research group of SLST published a research paper in Neuron entitled, “ASH1L haploinsufficiency results in autistic-like phenotypes in mice and links Eph receptor gene to autism spectrum disorder”. This study describes the role of Ash1l mutation in autism spectrum disorder (ASD), revealing that Ash1l loss-of-function resulted in ASD-like behavioral phenotypes and deficits in activity-induced synapse pruning.
Ash1l deficits might disturb activity-regulated refinement of the neural circuit to activate more unrelated neurons, which disrupted the fine-tuning of the neuronal network to reduce discrimination ability and contributed to ASD-like behaviors. Importantly, they have identified a molecule EphA7, whose expression was regulated by Ash1l, involved in Ash1l-mediated synapse elimination process and abnormal behaviors.
ASD is a complex developmental disorder characterized by social communication defect, restricted interests, stereotyped and repetitive behavior. Many studies suggested that some genetic factors, as well as environmental factors, might lead to ASD. However, the mechanisms underlying the pathogenesis of ASD remain elusive. Recent studies revealed some ASD associated genes. Mutations on epigenetic regulators have been identified as one of the key risk factors in determining the predisposition to autism. As a member of the trithorax group proteins, Ash1l encodes a histone methyltransferase.

Figure 1. (Top) April 2 is world autism awareness day, which reminds us to take care of autistic patients. (Bottom left) Autism is a complex developmental disorder characterized by many symptoms and causes. (Bottom right) In mice study, researchers usually use three-chamber test to evaluate social ability. (Pictures were modified from internet.)
Guan’s lab used two-photon microscope to directly observe the synapse changes in mouse brain. They found that Ash1l loss-of-function leads to the behavioral abnormalities in mice, including repetitive behavior, social communication defect, increased anxiety and discrimination deficits. Interestingly, they found that loss of Ash1l gene in forebrain neurons could result in the same ASD-associated behavioral defects, suggesting the neuronal Ash1l is an essential regulator of ASD-related behavior. In those abnormal mice, spine densities on cortical neurons were significantly increased. As the learning engaged synapse elimination is lost in the Ash1l mutant animal, the neurons showed excessive synaptic connections and widen tuning in behavior toward a spectrum of stimuli.
As to the mechanism of Ash1l-induced synapse and behavior deficits, Guan’s lab identified a downstream effector, Eph receptor A7 (EphA7), a synapse-related protein was reduced in the Ash1l mutant mice. Furthermore, they found that expression of EphA7 was regulated by Ash1l through antagonizing EZH2-H3K27me3 inhibition. Applying drugs targeting EZH2 or HDACs could rescue the expression of EphA7 in Ash1l mutant neurons. Importantly, they found that directly activating the remaining EphA7 in the mutant mice could rescue synapse pruning deficits in cultured neurons and alleviate some behavioral deficits in the Ash1l+/- mice. Thus, this study revealed the synaptic mechanisms underlying some behavioral deficient in ASD and provided potential targets to treat the Ash1l-associated ASD in the future.
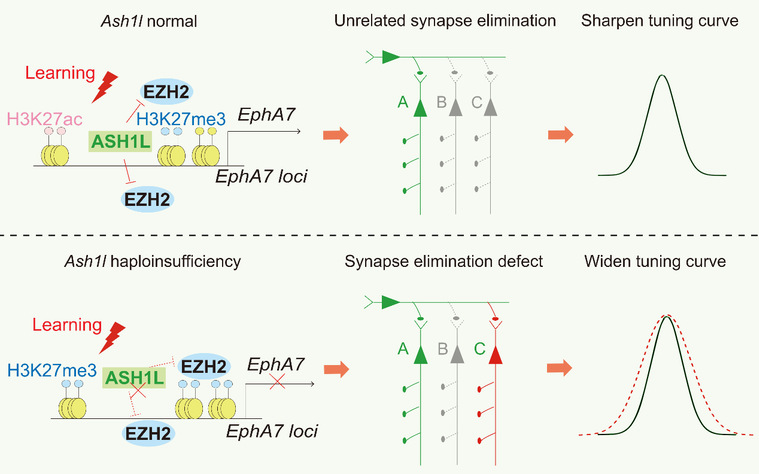
Figure 2. Schematic model illustrates that Ash1l regulates expression of EphA7 via antagonizing EZH2-H3K27me3 repression, preserves activity-dependent synapse elimination to avoid unrelated neurons activating, and sharpens tuning curve in discrimination performance.
Link to the article: https://www.cell.com/neuron/fulltext/S0896-6273(21)01090-4

