Mitochondria and peroxisomes are widely recognized as independent but functionally close-related organelles. Both are critical for the metabolism of lipids and reactive oxygen species. MARCH5 is a mitochondrion-associated ubiquitin ligase, with a function of regulating mitochondria fission and fusion, so as to maintain a dynamic balance of mitochondria. It also promotes mitophagy and regulates mitochondrial import. However, some of its roles seem controversial, suggesting other potential functions of MARCH5.
Recently, the research group led by Assistant Professor Zhuang Min from SLST published an article entitled “Ubiquitin ligase MARCH5 localizes to peroxisomes to regulate pexophagy” in Journal of Cell Biology. This study reveals that MARCH5 localizes to peroxisomes and regulates pexophagy via ubiquitinating PMP70. The study expands our knowledge about ubiquitin ligase MARCH5 and extends its functional analysis from mitochondria to peroxisomes.
In this study, the researchers used a proximity tagging system PUP-IT (developed by SLST Assistant Professor Zhuang Min, read more at: https://www.shanghaitech.edu.cn
/eng/2018/0815/c1418a31492/page.htm) to identify MARCH5 interacting proteins. Surprisingly, in addition to the known MARCH5 substrates on mitochondria, several peroxisomal proteins were identified that interact with MARCH5. They validated those interactions and further identified the role of MARCH5 in peroxisome degradation (pexophagy). In summary, there are two major findings in this study. One is identifying MARCH5 as a dual organelle locating protein targeted to the peroxisome by PEX19/PEX3. The second is the discovery that peroxisomal MARCH5 mediates PMP70 ubiquitination in Torin1 induced pexophagy.
In this paper, Ph.D. student of the Class of 2021 Zheng Jun and Research Assistant Professor Chen Xi from Zhuang’s lab are the co-first authors.
Link to the paper: https://doi.org/10.1083/jcb.202103156
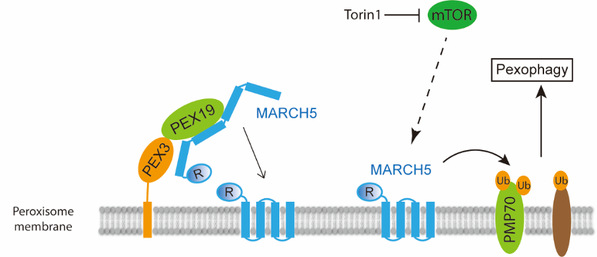
The working model for the localization and function of MARCH5 on peroxisomes

