Following myocardial infarction, the heart undergoes adverse remodeling driven by inflammation, cell necrosis, and fibrotic scar formation, ultimately leading to heart failure. However, the cellular and molecular mechanisms driving this process have not been fully elucidated.
Recently, a research team led by Associate Professor Zhang Hui of the School of Life Science and Technology (SLST) at ShanghaiTech University, in collaboration with Associate Professor Tang Juan from Tongji University, published a new study in Circulation. This study systematically reveals, for the first time, the critical role of P16+ cells in adverse cardiac remodeling and proposes a novel precision intervention strategy targeting CCL8 signaling or P16+ fibroblasts. These findings provide an important theoretical foundation for developing new myocardial infarction drugs based on immune modulation and precision cell therapy.

The team utilized cell lineage tracing technology to label cells, creating the identity atlas of P16+ cells in the heart after myocardial infarction. The study found that within the damaged cardiac region, fibroblasts, macrophages, vascular endothelial cells, and cardiomyocytes, all showed high expression of P16+. The team pinpointed two key cell types—P16+ fibroblasts and macrophages—which are not mere bystanders but active “instigators” of the pathology. These two cell types abundantly secrete the chemokine CCL8, recruiting cytotoxic lymphocytes, particularly CD8+ killer T cells, to the damaged heart tissue. These recruited immune cells release cytotoxic molecules that directly induce cardiomyocyte apoptosis, further exacerbating structural disruption and cardiac dysfunction (Figure 1).
To validate this mechanism, the team conducted a series of intervention experiments. The results showed that neutralizing CCL8 with antibodies or conditionally knocking out the Ccl8 gene in P16+ cells using Cre/loxP technology significantly reduced cytotoxic lymphocyte infiltration, protected cardiomyocytes, diminished the post-infarction scar area, and effectively improved cardiac function (Figure 1).
Of particular note, the team employed a “dual-lock” genetic technology (a dual-recombinase-mediated diphtheria toxin receptor expression system) to achieve precise ablation of either P16+ fibroblasts or macrophages. The results revealed that ablating only P16+ fibroblasts significantly promoted cardiac repair, whereas ablating P16+ macrophages had no notable effect. This suggests that P16+ fibroblasts are the key cell subpopulation driving adverse remodeling and represent a precise target for future drug intervention.
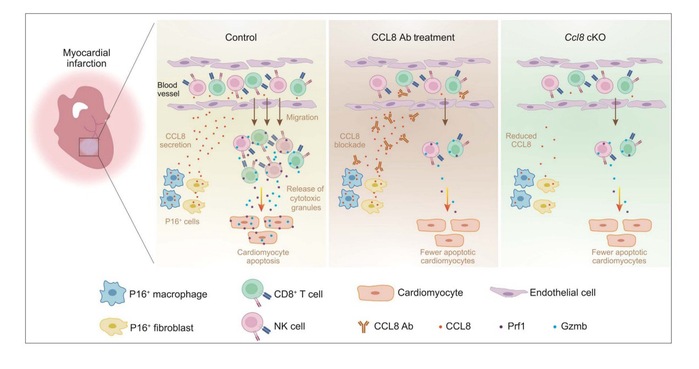
Figure 1. P16+ cells recruit CD8+ T cells and natural killer (NK) cells via CCL8 secretion, inducing cardiomyocyte apoptosis and driving adverse cardiac remodeling after myocardial infarction.
“In the past, we only knew that P16+ cells were associated with aging, but their specific function in heart disease remained largely unknown,” said Prof. Zhang. “This study not only clarifies their identity but also reveals how they collaborate with the immune system to exacerbate injury. This suggests that treating myocardial infarction may not require the elimination of all senescent cells. By blocking the CCL8 signaling pathway or precisely ablating P16+ fibroblasts, we aim to minimize collateral damage to normal cells during intervention, thereby achieving safer and more effective therapies.”
The co-first authors of this paper are Yan Lei and Zheng Jialei, doctoral students from the SLST at ShanghaiTech University, and Lu Zhengkai, a postdoctoral researcher at Tongji University. Profs. Zhang Hui and Tang Juan are the co-corresponding authors. ShanghaiTech University is the first affiliation.
*This article is provided by Prof. Zhang Hui

