Our body has special immune cells called T cells that fight tumors. On the surface of T cells is a protein called TCR, which recognizes a small special marker (antigen) on the surface of tumor cells. Once it detects the marker, the T cell launches an attack to eliminate the tumor.
However, many tumors escape detection by altering, reducing, or completely removing this marker, making it hard for T cells to recognize them. Scientists are working on ways to make TCR more sensitive—so it can trigger an attack even when very little antigen is present—while ensuring it does not mistakenly attack normal cells.
Previously, one common way to modify TCR was to increase its affinity to the antigen, making it bind very tightly. But this approach carries high risk—tumor antigens are often very similar to antigens on normal cells, and a TCR with too high affinity can easily recognize normal cells as targets, causing severe side effects (there have been real fatal cases in clinical trials).
Later research revealed that what really determines how strongly T cells activate is not the static affinity, but whether TCR and the antigen can hold on more tightly under molecular tensile force, a phenomenon called catch bond. When an appropriate tensile force is applied, which naturally occurs at the cell surface, the binding lasts longer instead of breaking quickly. This prolonged binding allows T cells to receive a stronger signal and activate more effectively. For incorrect or weak antigens, the force usually causes the binding to break rapidly, preventing mistaken activation.
Modifying this “hold on tighter under molecular tensile force” ability previously required precise 3D structure images of TCR and the antigen marker, which is difficult and often impossible to obtain.
Now, a joint research team led by Associate Professor Sun Bo from the School of Life Science and Technology (SLST) and Professor Zhao Xiang at the Center for Excellence in Molecular Cell Science at the Chinese Academy of Sciences, has found a simple approach. Their study was published in an article titled “Tuning the sensitivity of mechanosensory receptors through histidine scanning” in the journal Cell. In the research, they replaced individual amino acids in the key regions of TCR one by one with a common amino acid called histidine. After replacement, they tested which positions make T cells react more strongly, quickly identifying the effective spots.

They tested many different TCRs targeting viruses and various tumors, and for each one, they found about 10 good positions. This proves that histidine replacement works effectively for a wide range of TCRs.
Why does histidine work so well? Under molecular tensile force, it can form several additional hydrogen bonds and salt bridges with the antigen marker, making the binding last longer. Computer simulations and microscopic force experiments confirmed this. As a result, the internal signaling in T cells is amplified, leading to stronger activation.
The team carefully checked safety—these modified TCRs do not mistakenly recognize other markers and do not cause extra side effects. In laboratory and mouse experiments, T cells modified with this method treated melanoma, blood cancer, colorectal cancer, bone cancer, and others much more effectively than the original versions—better tumor control and longer survival times.
This method is not limited to T cells. It also works for other protein systems that rely on molecular tensile force such as Notch, which controls cell development, and Fc systems, which affect antibody functions, making them more sensitive.
In summary, scientists have developed a method that does not require complex structure images and only needs to replace one type of amino acid to make T cell recognition much more sensitive. It is safe, simple, and holds great promise for helping more cancer patients.
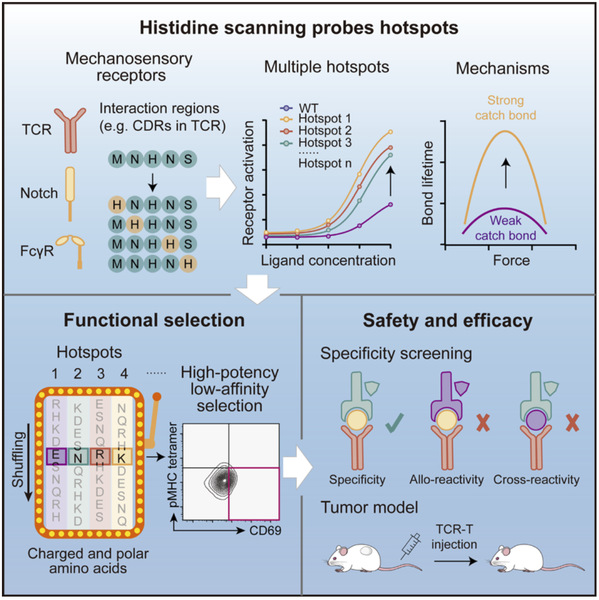
Simple replacement with histidine allows TCR to bind more persistently under molecular tensile force.
Prof. Sun Bo is one of the corresponding authors, and PhD student Bao Yanling from SLST is one of the first authors.

