On February 11, a research team led by Ge Jingpeng from the School of Life Science and Technology (SLST) at ShanghaiTech University and Yu Jie from the Interdisciplinary Research Center on Biology and Chemistry (IRCBC) at the Shanghai Institute of Organic Chemistry published a study in Nature entitled “Conformational diversity and fully opening mechanism of native NMDA receptor.”
The research team extracted native NMDA receptors directly from the whole mouse brain. Using immunoaffinity purification, single-molecule fluorescence microscopy, and cryo-electron microscopy, they obtained clear images of 10 different receptor structures. These structures show the various compositions and conformations that these receptors take in the brain and, for the first time, capture the fully open state of the receptor.
NMDA receptors are important proteins in the brain that help nerve cells send signals to each other. They play a key role in learning and memory. When these receptors do not work normally, they can lead to diseases such as epilepsy, schizophrenia, depression, or Alzheimer’s disease.
NMDA receptors are made up of four subunits, usually two GluN1 subunits plus two other subunits (such as GluN2A or GluN2B). Different combinations make the receptors open at different speeds and stay open for different lengths of time—receptors with GluN2A open quickly, while those with GluN2B open more slowly but stay open longer. Understanding the real shapes and opening ways of these receptors in the brain is important for developing drugs that target specific diseases in the future.
Most previous structural studies used receptors expressed in recombinant cultured cell lines in the laboratory. Although they provided a lot of useful information, these receptors may differ from the real ones in the brain in terms of their surrounding environment and subunit combinations. Therefore, directly extracting and observing native receptors from brain tissue has always been a major challenge.
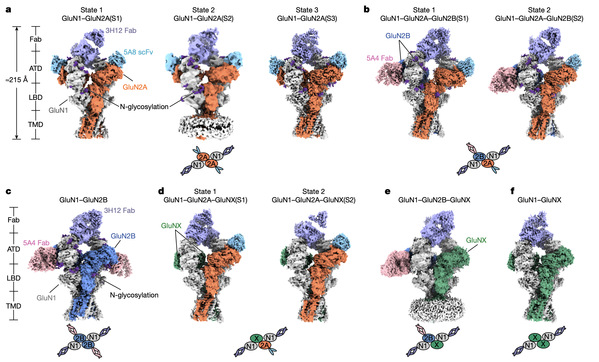
Ten assemblies of native NMDA receptors and their subunit compositions.
This study found that in the whole brain, receptors containing the GluN2A subunit are the most common subtypes. The amino-terminal domain (ATD) of GluN2A shows large changes in shape. These changes affect signal transmission from the top to the bottom of the receptor and make the receptor open faster. Combined with other experiments, the results show that these shape changes in the ATD are an important reason why receptors with GluN2A open quickly and have special responses to drugs.
Of particular importance, the team captured the fully open state in a native GluN1–GluN2B receptor. In this state, two key parts of the receptor channel both rotate outward, making the channel hole much larger so that ions can pass through easily. This is different from the only partially open state seen in earlier recombinantly expressed receptors and represents the true fully open state. It provides direct evidence for how ligand binding leads to complete channel opening.
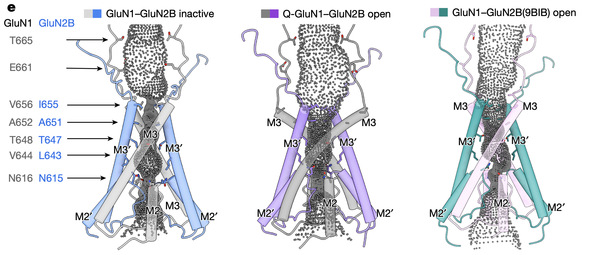
Pore diameter distribution of the native GluN1–GluN2B receptor.
(Left) Non-active (inactive/closed) state. (Middle) Fully open state. (Right) Previously reported open state.
Together, these findings reveal the multiple compositions and opening mechanisms of native NMDA receptors in the brain. They confirm that GluN2A plays the main role in receptor assembly across the whole brain and provide basic structural information for future studies of receptor function in the brain.
Xu Ruisheng, a PhD student from the IRCBC is the first author. Jiang Qiqi, a master’s student from SLST and Shanghai Clinical Research and Trial Center is the second author. Professors Yu Jie and Ge Jingpeng are the co-corresponding authors.

